Arcoxia
By W. Sven. Wartburg College. 2018.
The committee believes that the real or perceived mixing of public health and intellectual property concerns only holds back action on the problem of falsifed and substandard drugs purchase arcoxia 120mg line arthritis pain medication names. In her opening remarks, Director-General Margaret Chan reiterated the organization’s commitment to working against harmful products in the drug supply and promoting the availability of good-quality medicines around the world (Chan, 2012). Because the problem has legal dimensions, it will also be crucial to include experts in law enforcement, criminal justice, and customs. Contributing to the law enforcement and criminal justice sections of an international code on falsifed and substandard medi- cines would draw on the agency’s strengths and complement the goals set out in its 3-year strategy. National customs offces are under pressure to facilitate international trade and to monitor the safety of products enter- ing the country; they have a unique understanding of the circumstances through which illegitimate medicines enter commerce. Monitoring the trade in illegitimate medicines and enforcing laws against them depend on customs bureaus, however. Failing to include them in the development of the code would risk its being unacceptable or impractical for customs offcers, one of the main groups that would need to adhere to it. Unicef continues to work with legislators and lawyers to implement maternity protection laws in more countries (Unicef, 2012). Recommendation 7-1: The World Health Assembly, in partnership with the United Nations Offce on Drugs and Crime and the World Customs Organization, and in consultation with major stakeholders, should institute an inclusive, transparent process for developing a code of practice on the global problem of falsifed and substandard medicines. The code should include guidelines on surveillance, regulation, and law enforcement, empowering states and the international community to prevent and respond to drug quality problems. At a minimum, however, the committee recommends that the process give some attention to interna- tional surveillance, drug regulation, and law enforcement as main areas in which to give guidance. International Surveillance As Chapter 3 explains, surveillance for substandard and falsifed drugs is uncoordinated, largely voluntary, and highly variable. The modern drug Key Findings and Conclusions • The international surveillance component of the code of practice should provide guidelines on how to develop a surveillance system for falsifed and substandard drugs and how to link it to routine pharmacovigilance. International surveillance is necessary to de- fne the magnitude of the problem and to identify priority areas for action. The sections of the code that discuss surveillance should give guidance on how to set up routine drug quality surveillance and how to make stra- tegic choices about which drugs to monitor in the most vulnerable regions. Once routine surveillance systems are running, data gleaned from them will inform some of these choices in an iterative process. It may be necessary to use active surveillance methods for some high-risk drugs and passive surveillance for others. The code might also recommend how to choose and manage key sentinel surveillance sites. The guidelines should also explain how to tie monitoring for falsifed and substandard drugs to routine phar- macovigilance and how to link surveillance with response. And, as Chapter 6 explains, these assays are expensive; running even mini- mal tests could quickly bankrupt a small county’s annual drug testing bud- get. The code should suggest ways to accommodate the added burden that surveillance will place on drug quality laboratories. There may be room for universities to take on more testing or for donors to fund dedicated, regional drug surveillance laboratories. The use of minilabs and hand-held detection technologies could also alleviate the added strain surveillance testing will place on drug quality laboratories. Building surveillance also requires building a workforce dedicated to data analysis and the prompt dissemination of public alerts when necessary. Therefore, using surveillance data effectively requires a strong medicines regulatory system. Guidelines on surveillance for falsifed and substan- dard drugs will depend on commensurate guidelines for the regulation of medicines. Medicines Regulation The proposed code of practice should give guidelines on the quality, safety, and effcacy of medicines that all countries can work toward. The code could suggest national minimum standards for licensing of importers, distributors, and wholesalers and guidelines on retail and dispensing of medicines. The code should direct countries to enact comprehensive medicines legislation that provides for all the drug regulatory functions, including the licensing of manufacturers and distributors, the issuing of market au- Copyright © National Academy of Sciences.
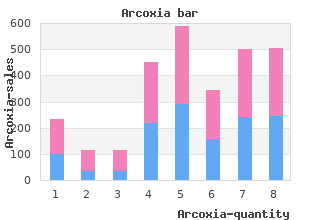
Taylor and colleagues collected 581 drugs from 35 randomly selected registered pharmacies in urban Nigeria (Taylor et al order arcoxia 120 mg mastercard rheumatoid arthritis lower back pain. They found 42 percent of antimalarials, 41 percent of antibacterials, and 54 percent of antituberculosis drugs outside of British Pharmacopoeia limits (Taylor et al. A stratifed random sample of medicine shops and licensed pharmacies in Laos found 90 percent of artesunate samples failed quality testing (Sengaloundeth et al. Researchers in southeast Nigeria attempted to include unlicensed private medicine dealers in their sample of antimalarial drug quality (Onwujekwe et al. They collected samples of a range of antimalari- als from patent medicine dealers, pharmacies, public and private hospitals, and primary health care centers (Onwujekwe et al. Pharmacopeia specifcations, by either not containing the active ingredient listed or containing it in low doses (Onwujekwe et al. Among the failed samples, 60 percent came from low-level shops, mostly the patent-medicine shops (Onwujekwe et al. Though most epidemiologically rigorous research on drug quality has tested antimicrobial drugs, there is some information about other essential medicines. In a 2012 study, Stanton and colleagues prepared an exhaus- tive sampling frame of formal and informal drug sellers in three districts in Ghana (Stanton et al. They chose 75 vendors at random from the sampling frame, from which patient actors collected 101 samples of ergometrine and oxytocin, the thermally unstable, uterotonic drugs used to treat postpartum hemorrhage (Stanton et al. A total of 89 percent of samples failed pharmacopeial testing; none of the ergometrine samples and only 26 percent of oxytocin samples met pharmacopeial specifcations (Stanton et al. All oxytocin samples (n = 46) were from unregistered manufacturers, though 18 were from manufacturers with registration pend- ing; 69 percent of ergometrine samples (n = 38) came from unregistered manufacturers, though 11 were from manufacturers with registration pend- ing (Stanton et al. A Need for More Field Surveys The best estimates of the scope of the drug supply affected come from systematic, random sampling and testing of medicines drawn from a rep- Copyright © National Academy of Sciences. The expense of required assays, discussed further in Chapter 6, is one barrier, but a large part of the problem is logistical. The frst step in drawing a systematic random sample of drugs is identifying the sampling frame, the list of every drug vendor in a given area. In developed coun- tries, registered pharmacies and dispensaries are the only place most of the population gets medicine. In low- and middle-income countries, however, there is often an extensive pharmaceutical gray market. Identifying all the vendors is diffcult and can be further complicated by the blurry lines be- tween licit and illicit commerce (Seear et al. Health workers may supplement their incomes by selling medicine informally (Peters and Bloom, 2012); peddlers may trade medicines occasionally, along with any number of dry goods, at bazaars and fea markets. Without formative research to catalogue the sampling frame, research on medicines quality is vulnerable to bias. Samples should be bought by patient actors, local study staff posing as shoppers who conceal from the vendor that they are working on an epidemiological investigation. Without taking steps to protect study validity, the researchers risk wast- ing time and money on a study that does not produce reliable estimates. For example, in 2009 the Indian government conducted a massive survey of drug quality across the country, estimating that only 0. Questions about the methodological rigor of the survey, particularly the choice of sampling frame and methods for sample collection, have called these results into question both within India and internationally (Bate, 2009, 2010; Pandeya, 2009). The committee supports the guidelines on feld surveys of medicine quality that Newton and colleagues proposed in March 2009 (Newton et al. They provide a standard protocol for collecting medicines samples and concrete advice on sampling techniques (Newton et al. More research adhering to the checklist in Table 3-8 would allow for a better understanding of the burden of falsifed and substandard drugs, and it would facilitate valid comparisons of the problem among countries and over time. There is no substitute, however, for pharmacovigilance and postmarket surveillance. It is not a coincidence that falsifed and substandard medicines circulate Copyright © National Academy of Sciences.

Adverse Effects Tachyphylaxis: flushing cheap arcoxia 60 mg otc arthritis hands medication, pruritus, and headache, which are mediated through prostaglandins and can be treated or prevented in some cases with antihistamines and/or aspirin; usually resolves over 1 to 2 weeks. Flushing is worse if niacin is taken with alcohol or hot beverages Hepatic: dose-related hepatotoxicity, particularly at more than 2g/d, and with over-the-counter preparations, including liver failure with sus- tained release. Mechanism of Action Omega-3 fatty acids are found in fatty fish (tuna, salmon, and swordfish), omega-3 fatty acids seem to protect against thrombosis, arrhyth- mia,inflammation, and hypertension, and improve endothelial function. Dosing Adolescents: 1000 mg/day Adults: 4 g/day or more Pharmacokinetics Response should be seen within 2 months. Summary Lifestyle modification is the primary treatment modality for lipid disorders, however, pharmacotherapy should be considered in children with high choles- terol and concerning risk factors. However, there are gaps in our knowledge regarding long-term safety and efficacy, and each child should be considered in the context of the family’s risk and attitudes toward pharmacotherapy. Improved understand- ing of the initiating factors of atherosclerosis and the noninvasive measurement of preclinical disease will allow us to target treatment more precisely and inter- pret lipid values more effectively in the context of other risk factors, which are, in reality, surrogate markers for risk. Effects of an ad libitum low-glycemic load diet on cardiovascular disease risk factors in obese young adults. The rela- tionship of reduction in incidence of coronary heart disease to cholesterol lowering. Acceptability and compliance with two forms of cholestyramine in the treatment of hypercholesterolemia in chil- dren: a randomized, crossover trial. A randomized crossover trial of combination pharmacologic therapy in children with familial hyperlipidemia. Evidence from randomized tri- als of pravastatin, simvastatin, and atorvastatin for cardiovascular disease prevention. Efficacy and safety of statin therapy in children with familial hypercholesterolemia: a randomized, double-blind, placebo-controlled trial with simvastatin. Efficacy and safety of lovastatin in adolescent males with heterozygous familial hypercholesterolemia: a randomized controlled trial. Treat- ment of familial hypercholesterolemia in children and adolescents: effect of 13. Efficacy and safety of statin therapy in children with familial hypercholesterolemia: a rand- omized controlled trial. Short-term efficacy and safety of pravastatin in 72 children with familial hypercholesterolemia. Inhibition of cholesterol synthesis by atorvastatin in homozygous familial hypercholesterolaemia. Efficacy and safety of atorvastatin in children and adolescents with familial hypercholesterolemia or severe hyperlipidemia: a multi- center, randomized, placebo-controlled trial. Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Pravastatin has cholesterol-low- ering independent effects on the artery wall of atherosclerotic monkeys. Effects of a low-carbohydrate diet on weight loss and cardiovascular risk factor in overweight adolescents. Synthesis and absorption markers of cholesterol in serum and lipoproteins during a large dose of statin treatment. Comparative efficacy and safety of pravastatin and cholestyramine alone and com- bined in patients with hypercholesterolemia. Comparative safety of atorvastatin 80 mg versus 10 mg derived from analysis of 49 completed trials in 14,236 patients. Incidence of hospitalized rhabdomyolysis in patients treated with lipid-lowering drugs. Efficacy and safety of ezetimibe added to ongoing statin therapy for treatment of patients with primary hypercholesterolemia. Efficacy and safety of a potent new selective cholesterol absorption inhibitor, ezetimibe, in patients with primary hypercholesterolemia. The effects of gemfibrozil on hyperlipidemia in children with persistent nephritic syndrome. Niacin decreases removal of high-density lipoprotein apolipoprotein A-I but not cholesterol ester by Hep G2 cells.
It contains estrone arcoxia 120 mg on line arthritis diet in hindi, equilin, and 17 (alpha)- methyl stearate, benzyl alcohol, sodium lauryl sulfate, dihydroequilin, together with smaller amounts of 17 glycerin, and mineral oil. Cyanocobalamin gel for intranasal administration is equivalent vitamin B12 activity. It is solution of cyanocobalamin with methylcellulose, sodium very hygroscopic in the anhydrous form and sparingly to citrate, citric acid, glycerin, and benzalkonium chloride in moderately soluble in water (1:80). After initial priming, each metered gel delivers an oxidizing or reducing agents (vitamin C), but not by auto- average of 500 mcg of cyanocobalamin, and the 2. Prepare a dispersion of item 1 in balance of item 3 in a separate vessel and add to step 2. Each gram of synthetic nonfluorinated corticosteroid, for topical derma- lotion contains 0. The corticosteroids constitute a class of pri- lauryl sulfate, light mineral oil, cetyl alcohol, stearyl alco- marily synthetic steroids used topically as anti-inflamma- hol, propylene glycol, methylparaben, propylparaben, sor- tory and antipruritic agents. Dexamethasone Sodium Phosphate Ointment Dexamethasone sodium phosphate is 9-fluoro-11(beta), topical steroid ointment containing dexamethasone sodium 17-dihydroxy-16(alpha)-methyl-21-(phosphonooxy)pre- phosphate equivalent to 0. Inactive ingredients are white thalmic ointment dexamethasone sodium phosphate is a petrolatum and mineral oil. Dexpanthenol Cream Bill of Materials Scale (g/100 g) Item Material Name Quantity/kg (g) 5. Add items 11 and 12 and mix (without vacuum) and item 9 and heat to 70°C and mix for 10 and cool down to 25°C. In a separate container add and dissolve items 7 and 8 in item 6 at 70°C and add to step 2. Cool to room temperature, stirring continuously water, add liquid paraffin, and stir, heating to until the air bubbles disappear. Diclofenac Diethylamine Gel Bill of Materials Scale (g/100 g) Item Material Name Quantity/kg (g) 1. Charge 90% of item 11 in a mixing vessel, heat and then add to step 5 and mix for 10 minutes, to 80°C; stir to produce vortex and add item 2 7. Dissolve item 1 in items 3 and 4 separately and to disperse after passing through 1-mm sieve; transfer to step 6 through a cloth filter; mix for mix for 5 minutes, avoiding foam. Formulations of Semisolid Drugs 147 Diclofenac Diethylammonium Gel Bill of Materials Scale (mg/g) Item Material Name Quantity/kg (g) q. Place water purified and alcohol in a 316 grade carbomer swells completely in the hydroalco- stainless steel mixing tank. Diclofenac Sodium Suppositories Bill of Materials Scale (mg/suppository) Item Material Name Quantity/1000 Suppositories (g) 12. Diclofenac Sodium Suppositories Bill of Materials Scale (mg/suppository) Item Material Name Quantity/1000 Suppositories (g) 50. Formulations of Semisolid Drugs 149 Diclofenac Sodium Suppositories Bill of Materials Scale (mg/suppository) Item Material Name Quantity/1000 Suppositories (g) 100. Dichlorobenzyl Alcohol Tooth Gel Bill of Materials Scale (g/100 g) Item Material Name Quantity/kg (g) 1. Dienestrol Vaginal Cream The active ingredient in dienestrol vaginal cream is dienes- acid, butylated hydroxyanisole, citric acid, sodium trol 0. Transfer molten fat at 70°C after passing vessel at 79°–75°C; hold molten fat at 70°C through a stainless steel filter to step above with continuous stirring at low speed. Homogenize at slow speed for 10 minutes; tem- add and dissolve parabens by stirring. In a separate vessel, take the balance of item 5 salicylate solution to the cream at 50°C while and sodium hydroxide pellets and sodium phos- stirring. Transfer step 3 to the paraben solution and mix for lavender oil, and glycerin at 40°C. Each diacetate in an emollient, occlusive base consisting of gram of cream contains 0.

